Chapter 39: Q. 6 (page 1136)
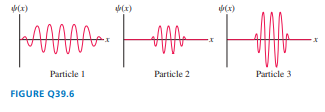
FIGURE Q39.6 shows wave packets for particles 1, 2, and 3. Which particle can have its velocity known most precisely? Explain.
Short Answer
The velocity of particle 1 is the most exact.
Learning Materials
Features
Discover
Chapter 39: Q. 6 (page 1136)
FIGURE Q39.6 shows wave packets for particles 1, 2, and 3. Which particle can have its velocity known most precisely? Explain.

The velocity of particle 1 is the most exact.
All the tools & learning materials you need for study success - in one app.
Get started for free
You are dealt 1 card each from 1000 decks of cards. What is the expected number of picture cards (jacks, queens, and kings)?
|You learned in Chapter 37 that, except for hydrogen, the mass of a nucleus with atomic number Z is larger than the mass of the Z protons. The additional mass was ultimately discovered to be due to neutrons, but prior to the discovery of the neutron it was suggested that a nucleus with mass number A might contain A protons and (A-Z) electrons. Such a nucleus would have the mase of A protone, but ite net charge would be only Z o.
a. We know that the diameter of a nuclens is approximately 10 fmm. Model the nucleus as a one-dimensional box and find the minimum range of speeds that an electron would have in such a box.
b. What does your answer imply about the possibility that the nucleus contains electrons? Explain.
The wave function of a particle is
where b is a positive constant. Find the probability that the particle is located in the interval -bx b
Consider a single-slit diffraction experiment using electrons. (Single-slit diffraction was described in Section 33.4.) Using Figure 39.5 as a model, draw
a. A dot picture showing the arrival positions of the first 40 or 50 electrons.
b. A graph of for the electrons on the detection screen.
c. A graph of for the electrons. Keep in mind that , as a wave-like function, oscillates between positive and negative.
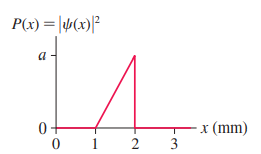
What is the value of the constant a in FIGURE Q39.5?
What do you think about this solution?
We value your feedback to improve our textbook solutions.