Chapter 20: Q. 50 (page 568)
FIGURE shows the thermal energy ofof gas as a function of temperature. What is for this gas?
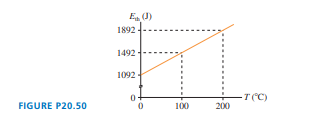
Short Answer
The for gas is
Learning Materials
Features
Discover
Chapter 20: Q. 50 (page 568)
FIGURE shows the thermal energy ofof gas as a function of temperature. What is for this gas?

The for gas is
All the tools & learning materials you need for study success - in one app.
Get started for free
The Nobel Prize in Physics was awarded for the discovery of graphene, a two-dimensional form of carbon in which the atoms form a two-dimensional crystal-lattice sheet only one atom thick. Predict the molar specific heat of graphene. Give your answer as a multiple of .
What are (a) the average kinetic energy and (b) the rms speed of a proton in the center of the sun, where the temperature is?
Eleven molecules have speeds . Calculate
(a) and
(b) .
For a monatomic gas, what is the ratio of the volume per atom to the volume of an atom when the mean free path is ten times the atomic diameter?
A mad engineer builds a cube,on a side, in which diameterrubber balls are constantly sent flying in random directions by vibrating walls. He will award a prize to anyone who can figure out how many balls are in the cube without entering it or taking out any of the balls. You decide to shoot diameter plastic balls into the cube, through a small hole, to see how far they get before colliding with a rubber ball. After many shots, you find they travel an average distance of . How many rubber balls do you think are in the cube?
What do you think about this solution?
We value your feedback to improve our textbook solutions.