Chapter 37: Q.11 (page 1081)
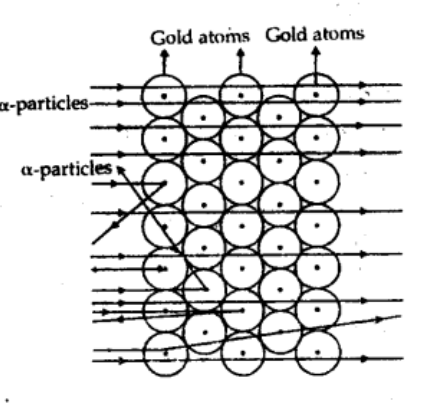
Explain why the observation of alpha particles scattered at very large angles led Rutherford to reject Thomson’s model of the atom and to propose a nuclear model.
Short Answer
Alpha particle scattered at very angles because when an alpha particle come very near to nucleus of gold atom then it feel high repulsive electric force in opposite direction and then it deflect with large angle