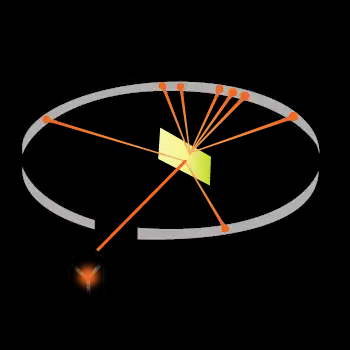
Rutherford passed beams of alpha particles through a thin gold foil and noted how the alpha particles scattered from the foil.
Most of the space inside the atom is empty because most of the α-particles passed through the gold foil without getting deflected.
Only a few particles were deflected from their path, indicating that the atom's positive charge takes up very little space.
A very small fraction of -particles was deflected by very large angles, indicating that the gold atom's positive charge and mass were concentrated in a very small volume within the atom.
Using X-rays, scientists later discovered the expected number of electrons (the same as the atomic number) in an atom. When an X-ray passes through an atom, some of it is scattered, while the rest is absorbed. Because X-rays lose their intensity primarily due to scattering at electrons, the number of electrons contained in an atom can be accurately estimated by noting the rate of decrease in X-ray intensity.
Alpha particles are scattered through large angles, because of close collisions with very massive and highly charged particles. Because the negatively and positively charged matter in this model is mixed together in a blob, any electric forces from this blog would mostly cancel out, resulting in only weak interaction forces for an alpha particle passing through a Thomson atom.




