Chapter 37: Q. 5 (page 1081)
Adiameter metal sphere is glowing red, but a spectrum shows that its emission spectrum peaks at an infrared wavelength of .How much power does the sphere radiate? Assume
Short Answer
The power that sphere radiates is
Learning Materials
Features
Discover
Chapter 37: Q. 5 (page 1081)
Adiameter metal sphere is glowing red, but a spectrum shows that its emission spectrum peaks at an infrared wavelength of .How much power does the sphere radiate? Assume
The power that sphere radiates is
All the tools & learning materials you need for study success - in one app.
Get started for free
4. What was the significance of Thomson’s experiment in which an off-center electrode was used to collect charge deflected by a magnetic field?
a. Summarize the experimental evidence prior to the research of Thomson by which you might conclude that cathode rays are some kind of particle.
b. Summarize the experimental evidence prior to the research of Thomson by which you might conclude that cathode rays are some kind of wave.
Determine:
a. The speed of a neutron.
b. The speed of a helium atom.
c. The specific type of particle that has of kinetic energy when moving with a speed of localid="1648723450518" role="math" .
FIGURE Q36.6 shows a rocket traveling from left to right. At the instant it is halfway between two trees, lightning simultaneously (in the rocket’s frame) hits both trees.
a. Do the light flashes reach the rocket pilot simultaneously? If not, which reaches her first? Explain.
b. A student was sitting on the ground halfway between the trees as the rocket passed overhead. According to the student, were the lightning strikes simultaneous? If not, which tree was hit first? Explain.
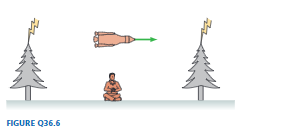
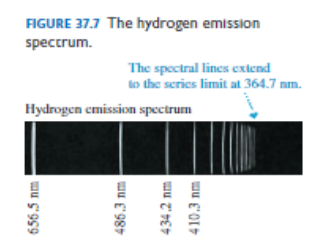
Figure 37.7 identified the wavelengths of four lines in the Balmer series of hydrogen.
a. Determine the Balmer formula n and m values for these wavelengths.
b. Predict the wavelength of the fifth line in the spectrum.
What do you think about this solution?
We value your feedback to improve our textbook solutions.