Chapter 14: 14.Q. 1 (page 383)
What is the volume in mL of 55 g of a liquid with density 1100 kg/m3?
Short Answer
Volume = 50 ml
Learning Materials
Features
Discover
Chapter 14: 14.Q. 1 (page 383)
What is the volume in mL of 55 g of a liquid with density 1100 kg/m3?
Volume = 50 ml
All the tools & learning materials you need for study success - in one app.
Get started for free
A 1.0-m-diameter vat of liquid is 2.0 m deep. The pressure at the bottom of the vat is 1.3 atm. What is the mass of the liquid in the vat?
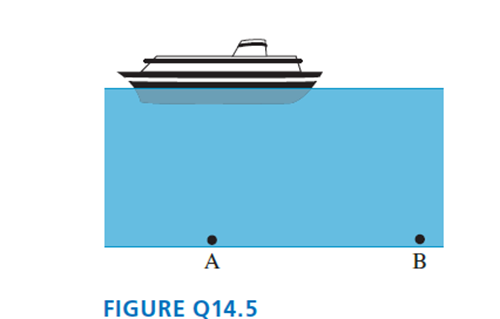
In FIGURE Q14.5, is pA larger than, smaller than, or equal to pB? Explain
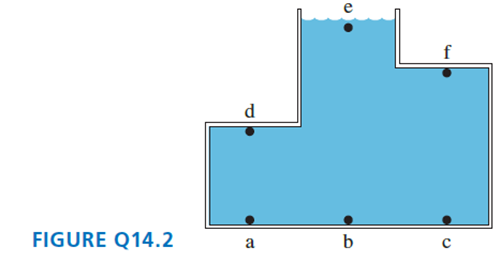
Rank in order, from largest to smallest, the pressures at a, b, and c in FIGURE Q14.2. Explain.
A spring with spring constant 35 N/m is attached to the ceiling, and a 5.0-cm-diameter, 1.0 kg metal cylinder is attached to its lower end. The cylinder is held so that the spring is neither stretched nor compressed, then a tank of water is placed underneath with the surface of the water just touching the bottom of the cylinder. When released, the cylinder will oscillate a few times but, damped by the water, quickly reach an equilibrium position. When in equilibrium, what length of the cylinder is submerged?
What is the volume in mL of 55 g of a liquid with density 1100 kg/m3?
What do you think about this solution?
We value your feedback to improve our textbook solutions.