Chapter 41: Q. 7 (page 1207)
What are and (as a multiple of) of a hydrogen atom in the state?
Short Answer
The hydrogen atom in the state isand.
Learning Materials
Features
Discover
Chapter 41: Q. 7 (page 1207)
What are and (as a multiple of) of a hydrogen atom in the state?
The hydrogen atom in the state isand.
All the tools & learning materials you need for study success - in one app.
Get started for free
Suppose you have a machine that gives you pieces of candy when you push a button. Eighty percent of the time, pushing the button gets you two pieces of candy. Twenty percent of the time, pushing the button yields pieces. The average number of pieces per push is . That is, pushes should get you, on average, pieces. Mathematically, the average value when the probabilities differ is . We can do the same thing in quantum mechanics, with the difference that the sum becomes an integral. If you measured the distance of the electron from the proton in many hydrogen atoms,
you would get many values, as indicated by the radial probability density. But the average value of would be
Calculate the average value of in terms of for the electron in the and the states of hydrogen.
For an electron in the state of hydrogen, what is the probability of being in a spherical shell of thickness at distance (a) , (b) , and (c) from the proton?
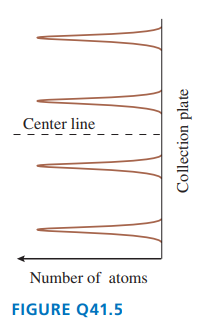
FIGURE Qshows the outcome of a Stern-Gerlach experiment with atoms of element .
a. Do the peaks represent different values of the atom’s total angular momentum or different values of the component of its angular momentum? Explain.
b. What angular momentum quantum numbers characterize these four peaks?
The transition in the emission spectrum of sodium has a wavelength of .What is the energy of thestate ?
a. Draw a diagram similar to Figure 41.2 to show all the possible orientations of the angular momentum vector for the case . Label each with the appropriate value of .
b. What is the minimum angle between and the z-axis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.