Chapter 41: Q. 14 (page 1207)
Show that .
Short Answer
The given statement is proved below.
Learning Materials
Features
Discover
Chapter 41: Q. 14 (page 1207)
Show that .
The given statement is proved below.
All the tools & learning materials you need for study success - in one app.
Get started for free
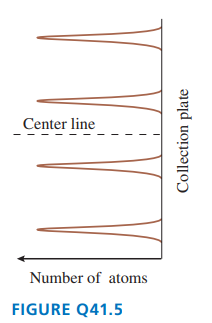
FIGURE Qshows the outcome of a Stern-Gerlach experiment with atoms of element .
a. Do the peaks represent different values of the atom’s total angular momentum or different values of the component of its angular momentum? Explain.
b. What angular momentum quantum numbers characterize these four peaks?
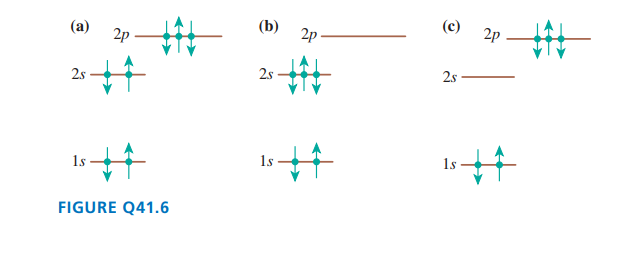
Does each of the configurations in FIGURE Qrepresent a possible electron configuration of an element? If so,
(i) identify the element and
(ii) determine whether this is the ground state or an excited state. If not, why not?
When all quantum numbers are considered, how many different quantum states are there for a hydrogen atom with? With? With? List the quantum numbers of each state.
Shows that the ionization energy of cadmiumis larger than that of its neighbors. Why is this?
Find the distance, in terms of , between the two peaks in the radial probability density of the state of hydrogen.
Hint: This problem requires a numerical solution.
What do you think about this solution?
We value your feedback to improve our textbook solutions.