Chapter 41: Q. 12 (page 1207)
Draw a series of pictures, similar to the ground states ofand.
Short Answer
The ground states of is , is,isandis.
Learning Materials
Features
Discover
Chapter 41: Q. 12 (page 1207)
Draw a series of pictures, similar to the ground states ofand.
The ground states of is , is,isandis.
All the tools & learning materials you need for study success - in one app.
Get started for free
sodium atoms are excited to the state at s. How many of these atoms remain in the state at
(a)
(b), and
(c) ?
Three electrons are in a one-dimensional rigid box (i.e., an infinite potential well) of length . Two are in the state and one is in the state. The selection rule for the rigid box allows only those transitions for which is odd.
a. Draw an energy-level diagram. On it, show the filled levels and show all transitions that could emit a photon.
b. What are all the possible wavelengths that could be emitted by this system?
The 1997 Nobel Prize in physics went to Steven Chu, Claude Cohen-Tannoudji, and William Phillips for their development of techniques to slow, stop, and “trap” atoms with laser light. To see how this works, consider a beam of rubidium atoms (mass ) traveling at after being evaporated out of an oven. A laser beam with a wavelength of is directed against the atoms. This is the wavelength of the transition in rubidium, with being the ground state, so the photons in the laser beam are easily absorbed by the atoms. After an average time of , an excited atom spontaneously emits a 780-nm-wavelength photon and returns to the ground state.
a. The energy-momentum-mass relationship of Einstein’s theory of relativity is . A photon is massless, so the momentum of a photon is . Assume that the atoms are traveling in the positive x-direction and the laser beam in the negative x-direction. What is the initial momentum of an atom leaving the oven? What is the momentum of a photon of light?
b. The total momentum of the atom and the photon must be conserved in the absorption processes. As a consequence, how many photons must be absorbed to bring the atom to a halt?
NOTE Momentum is also conserved in the emission processes. However, spontaneously emitted photons are emitted in random directions. Averaged over many absorption/emission cycles, the net recoil of the atom due to emission is zero and can be ignored.
c. Assume that the laser beam is so intense that a ground-state atom absorbs a photon instantly. How much time is required to stop the atoms?
d. Use Newton’s second law in the form to calculate the force exerted on the atoms by the photons. From this, calculate the atoms’ acceleration as they slow.
e. Over what distance is the beam of atoms brought to a halt?
What is the angular momentum of a hydrogen atom in
(a) a state and
(b) a state? Give your answers as a multiple of U.
shows the first few energy levels of the lithium atom. Make a table showing all the allowed transitions in the emission spectrum. For each transition, indicate
a. The wavelength, in nm.
b. Whether the transition is in the infrared, the visible, or the ultraviolet spectral region.
c. Whether or not the transition would be observed in the lithium absorption spectrum.
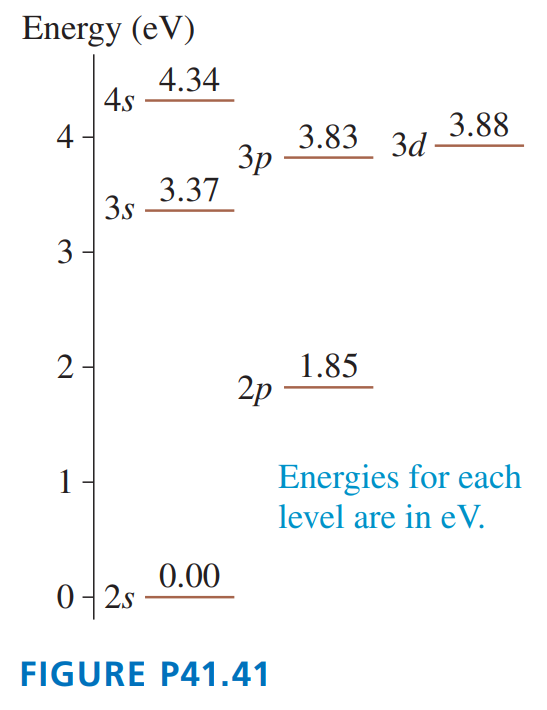
What do you think about this solution?
We value your feedback to improve our textbook solutions.