Chapter 18: Q. 42 (page 512)
The molecular mass of water (H2O) is A = 18. How many protons are there in 1.0 L of liquid water?
Short Answer
The answer is
Learning Materials
Features
Discover
Chapter 18: Q. 42 (page 512)
The molecular mass of water (H2O) is A = 18. How many protons are there in 1.0 L of liquid water?
The answer is
All the tools & learning materials you need for study success - in one app.
Get started for free
How many atoms are in a 2.0 cm * 2.0 cm * 2.0 cm cube of
aluminum?
A nebula—a region of the galaxy where new stars are forming—contains a very tenuous gas with . This gas is heated to by ultraviolet radiation from nearby stars. What is the gas pressure in atm?
An electric generating plant boils water to produce high-pressure steam. The steam spins a turbine that is connected to the generator.
a. How many liters of water must be boiled to fill a 5.0 m3 boiler with 50 atm of steam at 400C?
b. The steam has dropped to 2.0 atm pressure at 150C as it exits the turbine. How much volume does it now occupy?
To determine the mass of neon contained in a rigid, cylinder, you vary the cylinder’s temperature while recording the reading of a pressure gauge. Your data are as follows:
| Temperature (°C) | Pressure gauge (atm) |
| 100 | 6.52 |
| 150 | 7.80 |
| 200 | 8.83 |
| 250 | 9.59 |
Use the best-fit line of an appropriate graph to determine the mass of the neon.
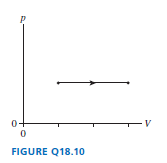
A gas under goes the process shown in FIGURE Q18.10. By what factor does the temperature change?
What do you think about this solution?
We value your feedback to improve our textbook solutions.