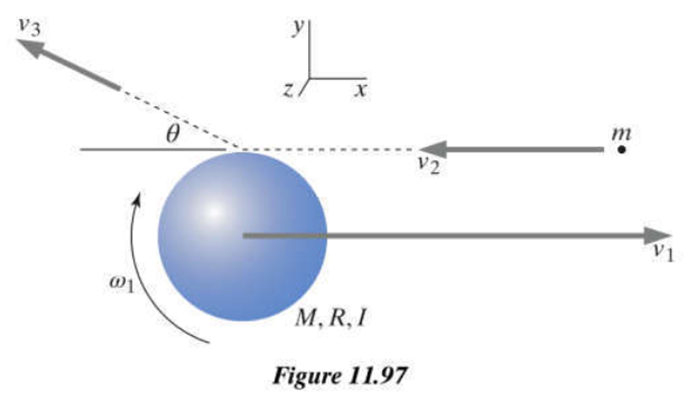
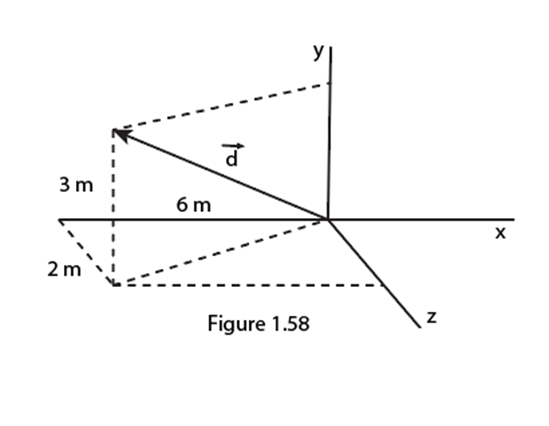
Chapter 2: Q62P (page 45)
A simplified model of a hydrogen atom is that the electron cloud is a sphere of radius with uniform charge density and total charge. (The actual charge density in the ground state is nonuniform.) See Figure 15.74.
a) For the uniform-density model, calculate the polarizability of atomic hydrogen in terms of . Consider the case where the magnitude of the applied electric field is much smaller than the electric field required to ionize the atom. Suggestions for your analysis: Imagine that the hydrogen atom is inside a capacitor whose uniform field polarizes but does not accelerate the atom. Consider forces on the proton in the equilibrium situation, where the proton is displaced a distance from the center of the electron cloud ( in the diagram). (b) For a hydrogen atom, can be taken as roughly (the Bohr model of the H atom gives ). Calculate a numerical value for the polarizability of atomic hydrogen. For comparison, the measured polarizability of a hydrogen atom is ; see the note below. (c) If the magnitude of the applied electric field is ,use the measured value of to calculate the shift shown in Figure 15.74. (d) For some purposes it is useful to model an atom as though the nucleus and electron cloud were connected by a spring. Use the measured value of to calculate the effective spring stiffness for atomic hydrogen. For comparison, measurements of Young’s modulus show that the effective spring stiffness of the interatomic force in solid aluminum is about . (e) If α were twice as large, what would be?
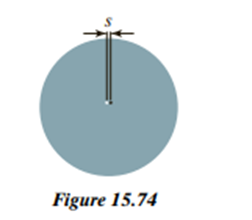
Note: Quantum-mechanical calculations agree with the experimental measurement of α reported in T. M. Miller and B. Bederson, “Atomic and molecular polarizabilities: a review of recent advances,” Advances in Atomic and Molecular Physics 13, 1–55, 1977. They use cgs units, so their value is 1/(4πε0 ) greater than the value given here.
Short Answer
(a) The expression for the polarization of atomic hydrogen in terms of is .
(b) The value of the polarization for the atomic hydrogen is and it is very close to measured polarization.
(c) The shift of the proton is .
(d) The value of the value of the effective string stiffness is .
(e) The value of the effective string stiffness for twice measured value of polarization is 173 N/m.