Chapter 8: Q6Q (page 344)
What is the energy of the photon emitted by the harmonic oscillator with stiffness and mass when it drops from energy level 5 to energy level 2?
Short Answer
The energy emitted by the photon is .
Learning Materials
Features
Discover
Chapter 8: Q6Q (page 344)
What is the energy of the photon emitted by the harmonic oscillator with stiffness and mass when it drops from energy level 5 to energy level 2?
The energy emitted by the photon is .
All the tools & learning materials you need for study success - in one app.
Get started for free
When starlight passes through a cold cloud of hydrogen gas, some hydrogen atoms absorb energy, then reradiate it in all directions. As a result, spectrum of the star shows dark absorption lines at the energies for which less energy from the star reaches us. How does the spectrum of dark absorption lines for very cold hydrogen differs from the spectrum of bright emission lines from very hot hydrogen?
The mean lifetime of a certain excited atomic state is 5 ns. What is the probability of the atom staying in this excited state for t=10 ns or more?
If you double the amplitude, what happens to the frequency in a classical (non quantum) harmonic oscillator? In a quantum harmonic oscillator?
A bottle contains a gas with atoms whose lowest four energy levels are , , , and . Electrons run through the bottle and excite the atoms so that at all times there are large numbers of atoms in each of these four energy levels, but there are no atoms in higher energy levels. List the energies of the photons that will be emitted by the gas.
Next, the electron beam is turned off, and all the atoms are in the ground state. Light containing a continuous spectrum of photon energies from to shines through the bottle. A photon detector on the other side of the bottle shows that some photon energies are depleted in the spectrum (“dark lines”). What are the energies of the missing photons?
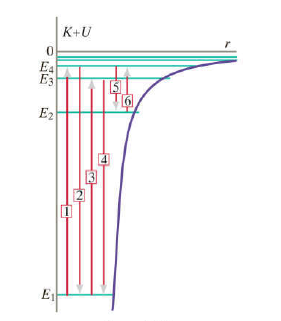
N=1 is the lowest electronic energy state for a hydrogen atom. (a) If a hydrogen atom is in a state N=4, what is K+U for this atom (in eV)? (b) The hydrogen atom makes a transition to state N=2, Now what is K+U in electron volts for this atom? (c) What is energy (in eV) of the photon emitted in the transition from level N=4 to N=2? (d) Which of the arrows in figure 8.40 represents this transition?
What do you think about this solution?
We value your feedback to improve our textbook solutions.