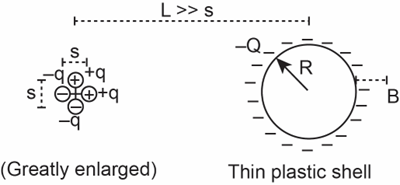
: A thin, hollow spherical plastic shell of radius \({\bf{R}}\)carries a uniformly distributed negative charge \({\bf{ - Q}}\). A slice through the plastic shell is shown in Figure 14.95. To the left of the spherical shell are four charges packed closely together as shown (the distance \({\bf{s}}\) is shown greatly enlarged for clarity). The distance from the center of the four charges to the center of the plastic shell is \({\bf{L}}\) , which is much larger than \({\bf{s}}\left( {{\bf{L}} \gg {\bf{s}}} \right)\). Remember that a uniformly charged sphere makes an electric field as though all the charge were concentrated at the center of the sphere.
(a)Calculate the \({\bf{x}}\) and \({\bf{y}}\) components of the electric field at location B, a distance \({\bf{b}}\) to the right of the outer surface of the plastic shell. Explain briefly, including showing the electric field on a diagram. Your results should not contain any symbols other than the given quantities \({\bf{R,Q,q,s,L}}\), and \({\bf{b}}\)(and fundamental constants). You need not simplify the final algebraic results except for taking into account the fact that \({\bf{L}} \gg {\bf{s}}\).
(b)What simplifying assumption did you have to make in part (a)?
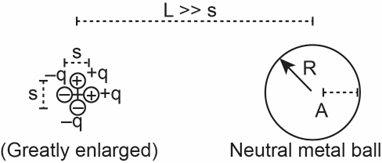
(c)The plastic shell is removed and replaced by an uncharged metal ball, as in Figure 14.96. At location Ainside the metal ball, a distance \({\bf{b}}\)to the left of the outer surface of the ball, accurately draw and label the electric field\({{\bf{\vec E}}_{{\bf{ball}}}}\) due to the ball charges and the electric field \({{\bf{\vec E}}_{\bf{4}}}\) of the four charges. Explain briefly.
(d)Show the distribution of ball charges.
(e)Calculate the \({\bf{x}}\) and \({\bf{y}}\) components of the net electric field at location A.