Chapter 19: Q73P (page 581)
At what frequency do molecules (diameter ) collide in (an ideal) oxygen gas ( ) at temperature and pressure ?
Short Answer
Frequency at which molecules collide in oxygen gas at and pressure is .
Learning Materials
Features
Discover
Chapter 19: Q73P (page 581)
At what frequency do molecules (diameter ) collide in (an ideal) oxygen gas ( ) at temperature and pressure ?
Frequency at which molecules collide in oxygen gas at and pressure is .
All the tools & learning materials you need for study success - in one app.
Get started for free
When the US submarine Squalus became disabled at a depth of, a cylindrical chamber was lowered from a ship to rescue the crew. The chamber had a radius ofand a height of, was open at the bottom, and held two rescuers. It slid along a guide cable that a diver had attached to a hatch on the submarine. Once the chamber reached the hatch and clamped to the hull, the crew could escape into the chamber. During the descent, air was released from tanks to prevent water from flooding the chamber. Assume that the interior air pressure matched the water pressure at depth h as given byrole="math" localid="1662369677002" , where
is the surface pressure andis the density of sea water.
Assume a surface temperature ofand a submerged water temperature of.
Question: From the knowledge that Cv , the molar specific heat at constant volume, for a gas in a container is 5.0 R , calculate the ratio of the speed of sound in that gas to the rms speed of the molecules, for gas temperature T. (Hint: See Problem 91.)
An ideal gas initially atis compressed at a constant pressure offrom a volume ofto a volume of. In the process,is lost by the gas as heat. What are
(a) the change in internal energy of the gas and
(b) the final temperature of the gas?
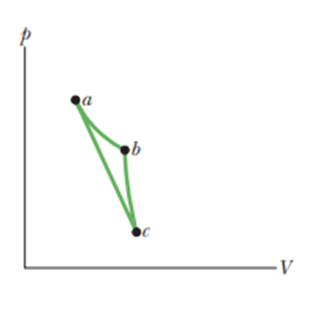
In the p-V diagram of Fig., the gas doesof work when taken along isotherm ab and when taken along adiabatic. What is the change in the internal energy of the gas when it is taken along the straight path from a to c?
Question: An air bubble of volume 20 cm3is at the bottom of a lake 40 mdeep, where the temperature is 4. 0 0C. The bubble rises to the surface, which is at a temperature of. Take the temperature of 20 0C the bubble’s air to be the same as that of the surrounding water. Just as the bubble reaches the surface, what is its volume?
What do you think about this solution?
We value your feedback to improve our textbook solutions.