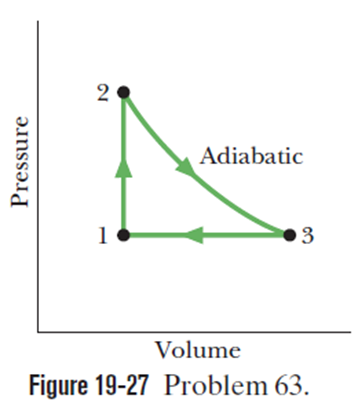
Chapter 19: Q94P (page 582)
Question: From the knowledge that Cv , the molar specific heat at constant volume, for a gas in a container is 5.0 R , calculate the ratio of the speed of sound in that gas to the rms speed of the molecules, for gas temperature T. (Hint: See Problem 91.)
Short Answer
Answer
The ratio of the speed of sound in the gas to the rms speed of the molecules of the gas, for the given temperature, T is 0.63.