Chapter 19: Q4Q (page 577)
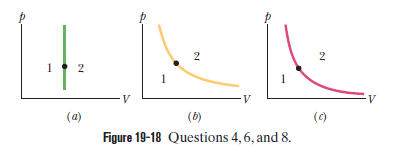
The dot in Fig 19-18 a represents the initial state of a gas, and the vertical line through the dot divides the p-V diagram into regions 1 and 2. For the following processes, determine whether the work W done by the gas is positive, negative, or zero: (a) the gas moves up along the vertical line, (b) it moves down along the vertical line, (c) it moves to anywhere in region 1, and (d) it moves to anywhere in region 2.
Short Answer
- Work done by the gas is if the gas moves up along the vertical line is zero.
- Work done by the gas is if the gas moves down along the vertical line is zero.
- Work done by the gas is if the gas moves anywhere in region 1 is negative.
- Work done by the gas is if the gas moves anywhere in region 2 is positive.