Chapter 19: Q50P (page 580)
We giveas heat to a diatomic gas, which then expands at constant pressure. The gas molecules rotate but do not oscillate. By how much does the internal energy of the gas increase?
Short Answer
.
Learning Materials
Features
Discover
Chapter 19: Q50P (page 580)
We giveas heat to a diatomic gas, which then expands at constant pressure. The gas molecules rotate but do not oscillate. By how much does the internal energy of the gas increase?
.
All the tools & learning materials you need for study success - in one app.
Get started for free
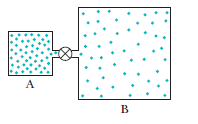
Question: Container A in figure holds an ideal gas at a pressure of and a temperature of 300 kIt is connected by a thin tube (and a closed valve) to container B, with four times the volume of A. Container B holds the same ideal gas at a pressure of and a temperature of 400 k. The valve is opened to allow the pressures to equalize, but the temperature of each container is maintained. What then is the pressure?
In the p-V diagram of Fig., the gas doesof work when taken along isotherm ab and when taken along adiabatic. What is the change in the internal energy of the gas when it is taken along the straight path from a to c?

One mole of an ideal diatomic gas goes from a to c along the diagonal path in figure. The scale of the vertical axis is set byand , and the scale of the horizontal axis is set by and. During the transition,
a) What is the change in internal energy of the gas
b) How much energy is added to the gas as heat?
c) How much heat is required if the gas goes from to along the indirect path ?
Suppose 12.0gof oxygen (O2) gas is heated at constant atmospheric pressure fromto.
Question: A container encloses 2 mol of an ideal gas that has molar mass M1 and 0.5 mol of a second ideal gas that has molar mass m2 = 3 .m1 What fraction of the total pressure on the container wall is attributable to the second gas? (The kinetic theory explanation of pressure leads to the experimentally discovered law of partial pressures for a mixture of gases that do not react chemically: The total pressure exerted by the mixture is equal to the sum of the pressures that the several gases would exert separately if each were to occupy the vessel alone.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.