Chapter 19: Q17P (page 578)
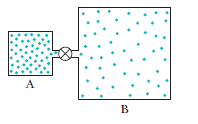
Question: Container A in figure holds an ideal gas at a pressure of and a temperature of 300 kIt is connected by a thin tube (and a closed valve) to container B, with four times the volume of A. Container B holds the same ideal gas at a pressure of and a temperature of 400 k. The valve is opened to allow the pressures to equalize, but the temperature of each container is maintained. What then is the pressure?
Short Answer
Answer
Pressure of the container after the valve is opened is.



