Chapter 19: Q59P (page 580)
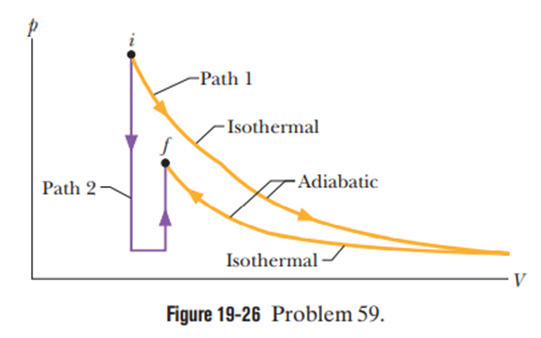
Figure shows two paths that may be taken by a gas from an initial point i to a final point f. Path 1 consists of an isothermal expansion (work is in magnitude), an adiabatic expansion (work is in magnitude), an isothermal compression (work is 30Jin magnitude) and then an adiabatic compression (work is 25Jin magnitude). What is the change in the internal energy of the gas if the gas goes from point i to point f along path 2?
Short Answer
The change in internal energy of the gas if gas goes from point i to f along path 2 is .