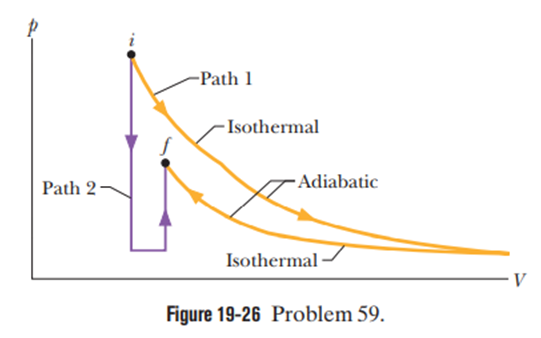
Chapter 19: Q57P (page 580)
The volume of an ideal gas is adiabatically reduced from 200Lto74.3L. The initial pressure and temperature are 1.00atmand 300K.The final pressure is 4.00atm.
- Is the gas monoatomic, diatomic or polyatomic?
- What is the final temperature?
- How many moles are in the gas?
Short Answer
- The gas is diatomic.
- The final temperature is Tf=446K.
- The number of moles in the gas is.