Chapter 19: Q72P (page 581)
At what temperature do atoms of helium gas have the same rms speed as molecules of hydrogen gas at ?
Short Answer
The temperature at which the atoms of helium gas have the same rms speed as molecules of hydrogen gas is .
Learning Materials
Features
Discover
Chapter 19: Q72P (page 581)
At what temperature do atoms of helium gas have the same rms speed as molecules of hydrogen gas at ?
The temperature at which the atoms of helium gas have the same rms speed as molecules of hydrogen gas is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: From the knowledge that Cv , the molar specific heat at constant volume, for a gas in a container is 5.0 R , calculate the ratio of the speed of sound in that gas to the rms speed of the molecules, for gas temperature T. (Hint: See Problem 91.)
During a compression at a constant pressure of , the volume of an ideal gas decreases from to . The initial temperature is , and the gas loses as heat.
Determine the average value of the translational kinetic energy of the molecules of an ideal gas at
a.
b.
What is the translational kinetic energy per mole of an ideal gas at
c.
d.
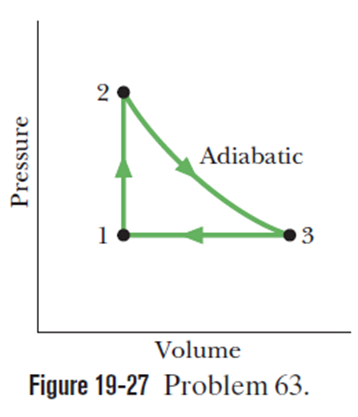
Figure shows a cycle undergone by 1.00molof an ideal monoatomic gas. The temperatures are T1=300K, T2=600K and T3=455K.For , what are- (a)Heat Q (b)The change in internal energy (c) The work done W? For, (d) What is Q? (e) What is Q? (f) What is W? For, (g)What is Q?(h) What is? (i) What is W? For the full cycle, (j) What is Q? (k)What is ? (l) What is W? The initial pressure at point 1 is 1.00atm.What are the (m) volume and (n) pressure at point 2? What are the (o) volume and (p) pressure at point 3?
Question: Find the rms speed of argon atoms at 313 K.Molar mass of argon atoms is 39.948 g/ mol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.