Chapter 19: Q21P (page 578)
Question: The lowest possible temperature in outer space is 2.7 K. What is the rms speed of hydrogen molecules at this temperature? (The molar mass is).
Short Answer
Answer:
The RMS speed of the hydrogen molecules is .
Learning Materials
Features
Discover
Chapter 19: Q21P (page 578)
Question: The lowest possible temperature in outer space is 2.7 K. What is the rms speed of hydrogen molecules at this temperature? (The molar mass is).
Answer:
The RMS speed of the hydrogen molecules is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Ten particles are moving with the following speeds: four at, two at, and four at. Calculate their
a) Average speed
b) Rms speed
c) Is?
The atmospheric density at an altitude of is about .
Question: From the knowledge that Cv , the molar specific heat at constant volume, for a gas in a container is 5.0 R , calculate the ratio of the speed of sound in that gas to the rms speed of the molecules, for gas temperature T. (Hint: See Problem 91.)
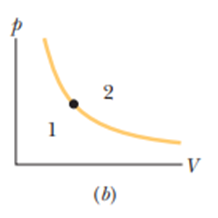
The dot in Figre 19-18bpresents the initial state of a gas, and the isotherm through the dot divides the p-V diagram into regions 1 and 2. For the following processes, determine whether the change in the internal energy of the gas is positive, negative, or zero: (a) the gas moves up along the isotherm, (b) it moves down along the isotherm, (c) it moves to anywhere in region, and (d) it moves to anywhere in region.
What do you think about this solution?
We value your feedback to improve our textbook solutions.