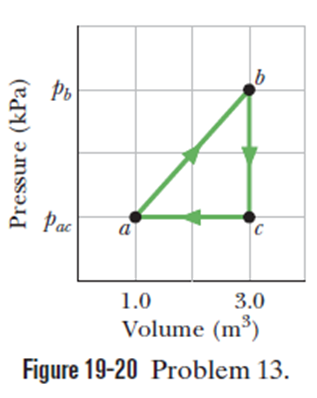
Chapter 19: Q6Q (page 577)
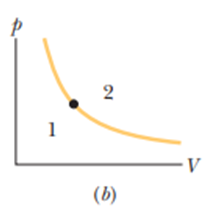
The dot in Figre 19-18bpresents the initial state of a gas, and the isotherm through the dot divides the p-V diagram into regions 1 and 2. For the following processes, determine whether the change in the internal energy of the gas is positive, negative, or zero: (a) the gas moves up along the isotherm, (b) it moves down along the isotherm, (c) it moves to anywhere in region, and (d) it moves to anywhere in region.
Short Answer
- The change in internal energy when the gas moves up along the isotherm is zero.
- The change in internal energy when the gas moves down along isotherm is zero.
- The change in internal energy when the gas moves to anywhere in region 1is negative.
- The change in internal energy when the gas moves to anywhere in region 2 is positive.