Chapter 19: Q70P (page 581)
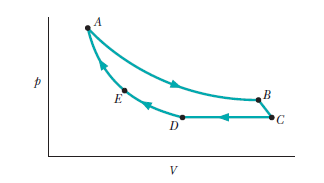
An ideal gas, at initial temperature and initial volume , is expanded adiabatically to a volume of , then expanded isothermally to a volume of , and then compressed adiabatically back to .What is its final volume?
Short Answer
The final volume, when the ideal gas again attains its initial temperature, is