Chapter 19: Q20P (page 578)
Question: Calculate the RMS speed of helium atoms at 1000k. Molar mass of helium atoms is.
Short Answer
Answer
The RMS speed of helium atoms at 1000 k is.
Learning Materials
Features
Discover
Chapter 19: Q20P (page 578)
Question: Calculate the RMS speed of helium atoms at 1000k. Molar mass of helium atoms is.
Answer
The RMS speed of helium atoms at 1000 k is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: An air bubble of volume 20 cm3is at the bottom of a lake 40 mdeep, where the temperature is 4. 0 0C. The bubble rises to the surface, which is at a temperature of. Take the temperature of 20 0C the bubble’s air to be the same as that of the surrounding water. Just as the bubble reaches the surface, what is its volume?
Question: A pipe of length L = 25. 0 m that is open at one end contains air at atmospheric pressure. It is thrust vertically into a freshwater lake until the water rises halfway up in the pipe (Fig.). What is the depth h of the lower end of the pipe? Assume that the temperature is the same everywhere and does not change.

A gas is to be expanded from initial state i to final state f along either path 1or path 2on a PV diagram. Path1 consists of three steps: an isothermal expansion (work isin magnitude), an adiabatic expansion (work isin magnitude), and another isothermal expansion (work isin magnitude). Path2 consists of two steps: a pressure reduction at constant volume and an expansion at constant pressure. What is the change in the internal energy of the gas along path 2?
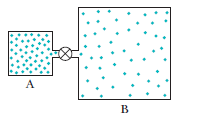
Question: Container A in figure holds an ideal gas at a pressure of and a temperature of 300 kIt is connected by a thin tube (and a closed valve) to container B, with four times the volume of A. Container B holds the same ideal gas at a pressure of and a temperature of 400 k. The valve is opened to allow the pressures to equalize, but the temperature of each container is maintained. What then is the pressure?
The envelope and basket of a hot-air balloon have a combined weight of , and the envelope has a capacity (volume) of . When it is fully inflated, what should be the temperature of the enclosed air to give the balloon a lifting capacity (force) of (in addition to the balloon’s weight)? Assume that the surrounding air, at , has a weight per unit volume of and a molecular mass of , and is at a pressure of .
What do you think about this solution?
We value your feedback to improve our textbook solutions.