Chapter 42: Q38P (page 1304)
A dose of of a radioactive isotope is injected into a patient. The isotope has a half-life of 3.0h. How many of the isotope parents are injected?
Short Answer
The number of injected isotope parents is .
Learning Materials
Features
Discover
Chapter 42: Q38P (page 1304)
A dose of of a radioactive isotope is injected into a patient. The isotope has a half-life of 3.0h. How many of the isotope parents are injected?
The number of injected isotope parents is .
All the tools & learning materials you need for study success - in one app.
Get started for free
What is the binding energy per nucleon of the americium isotope ? Here are some atomic masses and the neutron mass.
Consider a nucleus to be made up of an alpha particle () and a residual nucleus (). Plot the electrostatic potential energy U(r), where r is the distance between these particles. Cover the approximate range10 fm < r < 100 fm and compare your plot with that of Fig. 42-10.
In a certain rock, the ratio of lead atoms to uranium atoms is 0.300. Assume that uranium has a half-life ofand that the rock had no lead atoms when it formed. How old is the rock?
When aboveground nuclear tests were conducted, the explosions shot radioactive dust into the upper atmosphere. Global air circulations then spread the dust worldwide before it settled out on ground and water. One such test was conducted in October 1976. What fraction of the produced by that explosion still existed in October 2006? The half-life of .
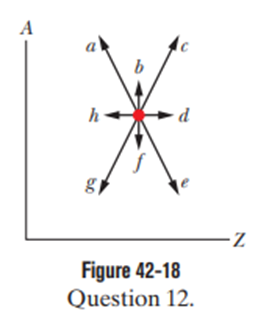
Figure 42-18 is a plot of mass number Aversus charge number Z.The location of a certain nucleus is represented by a dot. Which of the arrows extending from the dot would best represent the transition were the nucleus to undergo (a) adecay and (b) adecay?
What do you think about this solution?
We value your feedback to improve our textbook solutions.