Chapter 42: Q14P (page 1303)
What is the binding energy per nucleon of the americium isotope ? Here are some atomic masses and the neutron mass.
Short Answer
The binding energy per nucleon of the americium isotope is 7.52 MeV.
Learning Materials
Features
Discover
Chapter 42: Q14P (page 1303)
What is the binding energy per nucleon of the americium isotope ? Here are some atomic masses and the neutron mass.
The binding energy per nucleon of the americium isotope is 7.52 MeV.
All the tools & learning materials you need for study success - in one app.
Get started for free
At t=0, a sample of radionuclide Ahas the same decay rate as a sample of radionuclide Bhas at. The disintegration constants areand, with. Will the two samples ever have (simultaneously) the same decay rate? (Hint:Sketch a graph of their activities.)
The radionuclidedecays by emitting an electron. (a) Into which square in Fig. 42-6 is it transformed? (b) Do further decays then occur?
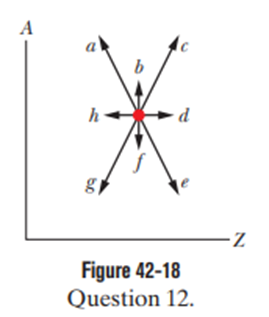
Figure 42-18 is a plot of mass number Aversus charge number Z.The location of a certain nucleus is represented by a dot. Which of the arrows extending from the dot would best represent the transition were the nucleus to undergo (a) adecay and (b) adecay?
nucleus with a kinetic energy of 3.00 MeV is sent toward a nucleus. What is the least center-to-center separation between the two nuclei, assuming that thenucleus does not move?
If the unit for atomic mass were defined so that the mass of were exactly 1.000 000 u, what would be the mass of(a) localid="1661600852143" (actual mass 12. 000 000 u ) localid="1661600855467" and (b) (actual mass 238.050 785 u)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.