Chapter 42: Q87P (page 1307)
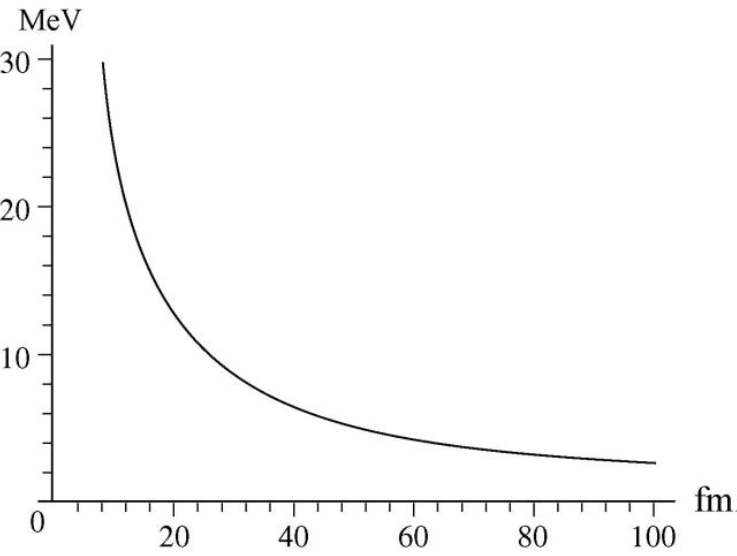
Consider a nucleus to be made up of an alpha particle () and a residual nucleus (). Plot the electrostatic potential energy U(r), where r is the distance between these particles. Cover the approximate range10 fm < r < 100 fm and compare your plot with that of Fig. 42-10.
Short Answer
The graph for electrostatic potential energy versus the distance between the particles is plotted.