Chapter 42: Q74P (page 1306)
In a certain rock, the ratio of lead atoms to uranium atoms is 0.300. Assume that uranium has a half-life ofand that the rock had no lead atoms when it formed. How old is the rock?
Short Answer
The rock is old.
Learning Materials
Features
Discover
Chapter 42: Q74P (page 1306)
In a certain rock, the ratio of lead atoms to uranium atoms is 0.300. Assume that uranium has a half-life ofand that the rock had no lead atoms when it formed. How old is the rock?
The rock is old.
All the tools & learning materials you need for study success - in one app.
Get started for free
Locate the nuclides displayed in Table 42-1 on the nuclidic chart of Fig. 42-5. Verify that they lie in the stability zone.
Cancer cells are more vulnerable to x and gamma radiation than are healthy cells. In the past, the standard source for radiation therapy was radioactive , which decays, with a half-life of 5.27y, into an excited nuclear state of.That nickel isotope then immediately emits two gamma-ray photons, each with an approximate energy of 1.2MeV. How many radioactivenuclei are present in a 6000Cisource of the type used in hospitals? (Energetic particles from linear accelerators are now used in radiation therapy.)
The radionuclide decays according to
The maximum energy of the emitted positrons is 0.960 MeV. (a) Show that the disintegration energy Qfor this process is given by
role="math" localid="1661759171201"
Whereandare the atomic masses ofand, respectively, andis the mass of a positron. (b) Given the mass values,and, calculate Qand compare it with the maximum energy of the emitted positron given above. (Hint:Let andbe the nuclear masses and then add in enough electrons to use the atomic masses.)
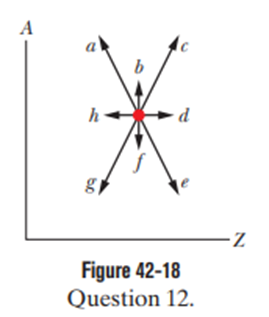
Figure 42-18 is a plot of mass number Aversus charge number Z.The location of a certain nucleus is represented by a dot. Which of the arrows extending from the dot would best represent the transition were the nucleus to undergo (a) adecay and (b) adecay?
An organic sample of mass4.00 kg absorbs2.00Jvia slow neutron radiation (RBE=5). What is the dose equivalent (mSv)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.