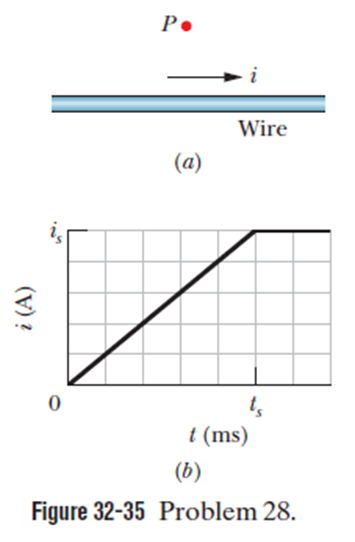
Chapter 32: Q33P (page 969)
If an electron in an atom has an orbital angular momentum with, what are the components (a) and (b)? If the atom is in an external magnetic fieldthat has magnitudeand is directed along the z axis, what are (c) the energyassociated withand(d) the energyassociated with? (e) If, instead, the electron has, what are (e)? (f) (g) and (h)?
Short Answer
a)Thecomponentis zero.
b) The component is zero
c) The energy associated with is zero
d)The energy associated with is
e)If, instead,the electron has , the value of is
f) If, instead, the electron has , the value of is
g) If, instead, the electron has , the value of is
h) If, instead, the electron has , the value of is