Chapter 41: Q28P (page 1274)
What is the Fermi energy of gold (a monovalent metal with molar mass 197 g/mol and density )?
Short Answer
The Fermi energy of gold is .
Learning Materials
Features
Discover
Chapter 41: Q28P (page 1274)
What is the Fermi energy of gold (a monovalent metal with molar mass 197 g/mol and density )?
The Fermi energy of gold is .
All the tools & learning materials you need for study success - in one app.
Get started for free
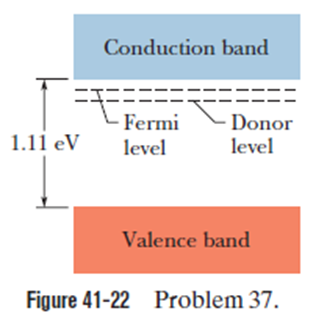
Doping changes the Fermi energy of a semiconductor. Consider silicon, with a gap of 1.11eV between the top of the valence band and the bottom of the conduction band. At 300K the Fermi level of the pure material is nearly at the mid-point of the gap. Suppose that silicon is doped with donor atoms, each of which has a state 0.15eV below the bottom of the silicon conduction band, and suppose further that doping raises the Fermi level to 0.11eV below the bottom of that band (Fig. 41-22). For (a) pure and (b) doped silicon, calculate the probability that a state at the bottom of the silicon conduction band is occupied. (c) Calculate the probability that a state in the doped material (at the donor level) is occupied.
In a simplified model of an undoped semiconductor, the actual distribution of energy states may be replaced by one in which there are states in the valence band, all having the same energy, andstates in the conduction band all these states having the same energy. The number of electrons in the conduction band equals the number of holes in the valence band.
(a) What maximum light wavelength will excite an electron in the valence band of diamond to the conduction band? The energy gap is 5.50 eV. (b) In what part of the electromagnetic spectrum does this wavelength lie?
Calculate the density of states for metal at energy and show that your result is consistent with the curve of Fig. 41-6.
What is the probability that, at a temperature of T = 300 K, an electron will jump across the energy gap in a diamond that has a mass equal to the mass of Earth? Use the molar mass of carbon in Appendix F; assume that in diamond there is one valence electron per carbon atom.
What do you think about this solution?
We value your feedback to improve our textbook solutions.