Chapter 4: Q. 4.19 (page 133)
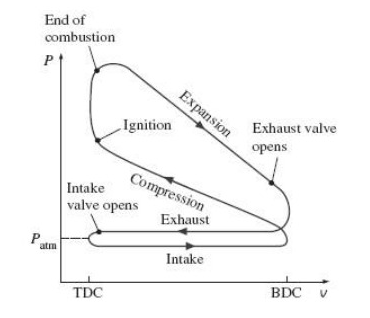
The amount of work done by each stroke of an automobile engine is controlled by the amount of fuel injected into the cylinder: the more fuel, the higher the temperature and pressure at points 3 and 4 in the cycle. But according to equation 4.10, the efficiency of the cycle depends only on the compression ratio (which is always the same for any particular engine), not on the amount of fuel consumed. Do you think this conclusion still holds when various other effects such as friction are taken into account? Would you expect a real engine to be most efficient when operating at high power or at low power? Explain.
Short Answer
The real engine is most efficient in low power than the high power.