Chapter 4: Q. 4.27 (page 137)
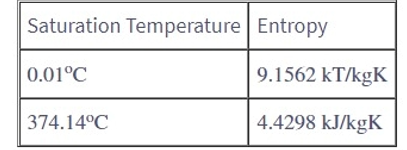
In table 4.1, why does the entropy of water increase with increasing temperature, while the entropy of steam decreases with increasing temperature?
Short Answer
Because the overall change in temperature outweighs the change in volume, entropy for water molecules rises.
In the case of steam, the overall change in volume drops more than the temperature increase, and so the entropy decreases.