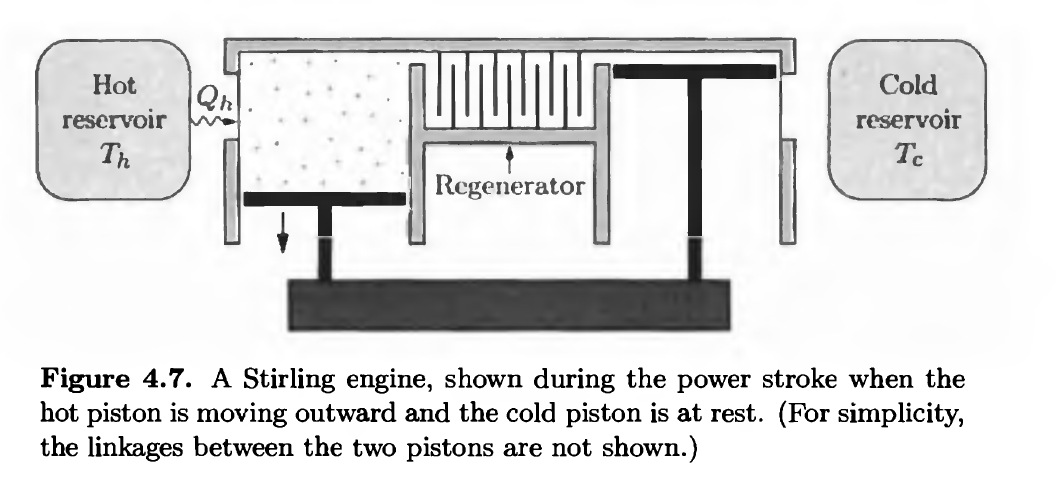
Chapter 4: Q.4.31 (page 141)
Suppose that the throttling valve in the refrigerator of the previous problem is replaced with a small turbine-generator in which the fluid expands adiabatically, doing work that contributes to powering the compressor. Will this change affect the COP of the refrigerator? If so, by how much? Why do you suppose real refrigerators use a throttle instead of a turbine?
Short Answer
The COP is enhanced by 1.08, and practical refrigerators use throttle rather than the turbine to reduce cost and complexity.