Chapter 1: Q. 1.33 (page 1)
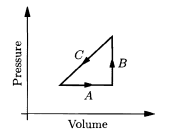
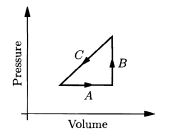
An ideal gas is made to undergo the cyclic process shown in the given figure. For each of the steps A, B, and C, determine whether each of the following is positive, negative, or zero: (a) the work done on the gas; (b) the change in the energy content of the gas; (c) the heat added to the gas.
Then determine the sign of each of these three quantities for the whole cycle. What does this process accomplish?
Short Answer
a. Total work done on the gas is
b. There is no net change in thermal energy.
c. Total heat added is .