Chapter 1: Q. 1.51 (page 36)
Use the data at the back of this book to determine for the combustion of a mole of glucose,
This is the (net) reaction that provides most of the energy needs in our bodies.
Short Answer
The value ofis.
Learning Materials
Features
Discover
Chapter 1: Q. 1.51 (page 36)
Use the data at the back of this book to determine for the combustion of a mole of glucose,
This is the (net) reaction that provides most of the energy needs in our bodies.
The value ofis.
All the tools & learning materials you need for study success - in one app.
Get started for free
Calculate the total thermal energy in a liter of helium at room temperature and atmospheric pressure. Then repeat the calculation for a liter of air.
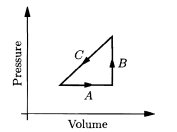
An ideal gas is made to undergo the cyclic process shown in the given figure. For each of the steps A, B, and C, determine whether each of the following is positive, negative, or zero: (a) the work done on the gas; (b) the change in the energy content of the gas; (c) the heat added to the gas.
Then determine the sign of each of these three quantities for the whole cycle. What does this process accomplish?
Pretend that you live in the th century and don't know the value of Avogadro's number* (or of Boltzmann's constant or of the mass or size of any molecule). Show how you could make a rough estimate of Avogadro's number from a measurement of the thermal conductivity of gas, together with other measurements that are relatively easy.
Uranium has two common isotopes, with atomic masses of 238 and 235. One way to separate these isotopes is to combine the uranium with fluorine to make uranium hexafluoride gas, UF6, then exploit the difference in the average thermal speeds of molecules containing the different isotopes. Calculate the rms speed of each type of molecule at room temperature, and compare them.
A battery is connected in series to a resistor, which is immersed in water (to prepare a nice hot cup of tea). Would you classify the flow of energy from the battery to the resistor as "heat" or "work"? What about the flow of energy from the resistor to the water?
What do you think about this solution?
We value your feedback to improve our textbook solutions.