To obtain work done on the system :
Here P is pressure, are initial volume and final volume.
At a pressure of , the volume of the water is reduced to of its value at atmospheric pressure .
Assume that the reduction in volume is linearly proportional to the increase in pressure.
The pressure is calculated as :
Here A and B are constant, P is pressure and V is volume.
At , the volume is
And at , the volume is
Let's take the standard temperature and pressure, , equation (2) can be written as by substitute
For equation (2) can be written as, by substituting
Solve equation (3) and (4) for the value of A and B
Now equation (1) can be written as
Work required to compress 1 liter of water to can be found by :
localid="1651133444001"
Substitute localid="1651133451713" for V to solve A and B
localid="1651133447933"
And similarly, for B, localid="1651133455574"
Substitute AV+B for P in equation (6)
localid="1651133459614"
Substitute localid="1651133463066"
localid="1651133466748"
If constant pressure of localid="1651133470929" is considered,
Work done will be localid="1651133474496"
Substitute localid="1651133477862"
localid="1651133481243"
Again substitute localid="1651133484260"
localid="1651133487776"
And the graph between pressure and volume is shown below which is followed by equation (5)
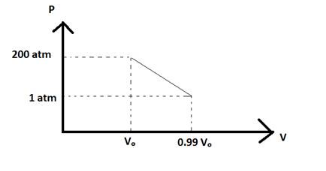
Hence, at constant pressure work done is and at variable pressure work done is


