Chapter 1: Q. 1.31P (page 22)
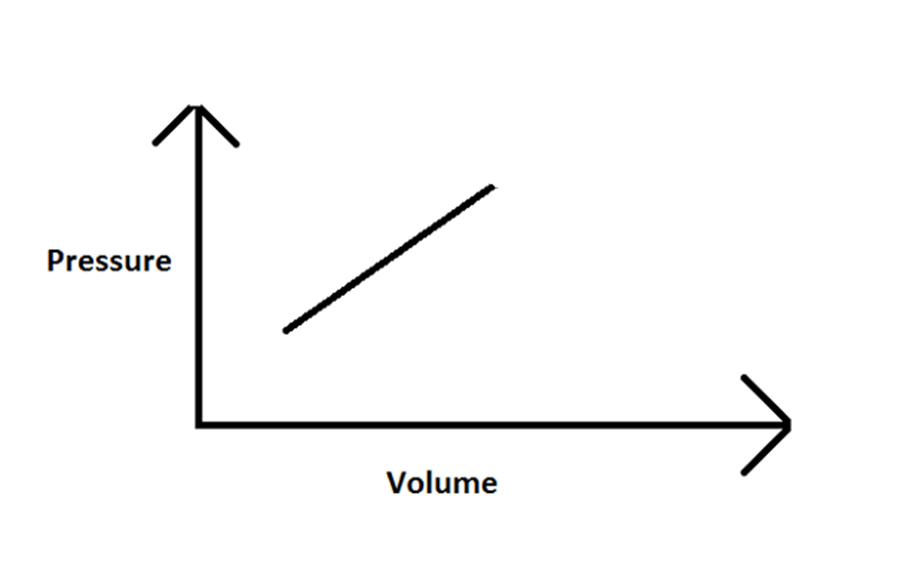
Imagine some helium in a cylinder with an initial volume of 1 liter and an initial pressure of 1 atm. Somehow the helium is made to expand to a final volume of 3 liters, in such a way that its pressure rises in direct proportion to its volume.
- Sketch a graph of pressure vs. volume for this process.
- Calculate the work done on the gas during this process, assuming that there are no “other” types of work being done.
- Calculate the change in the helium’s energy content during this process.
- Calculate the amount of heat added to or removed from the helium during this process.
- Describe what you might do to cause the pressure to rise as the helium expands.
Short Answer
- The graph between pressure and volume is a straight line but does not pass through the origin.
- Work done on the gas is .
- Change in helium’s energy is .
- The amount of heat added is .
- Pressure increases when kinetic energy increases. To increase pressure, kinetic should be increased and finally, it is concluded that one has to increase the temperature to increase pressure.