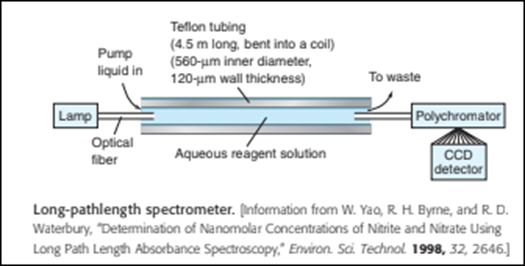
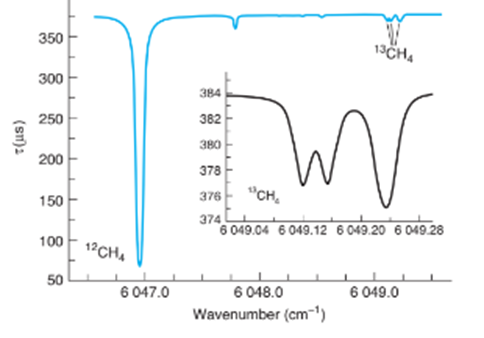
Chapter 20: Q8P (page 525)
Deuterated triglycine sulfate (abbreviated DTGS) is a common ferroelectric infrared detector material. Explain how it works.
Short Answer
Deuterated triglycine sulfate (DTGS) has a permanent electric polarization as a result of alignment of the molecules in the crystal. One part of the crystal is positively charged, while the opposite side is negative. The polarization is temperature dependent, so when the crystal absorbs infrared radiation, its temperature and polarization (voltage difference) change. The change in voltage is the detector pyroelectric signal.