Chapter 20: Q1P (page 525)
Describe the role of each component of the spectrophotometer.
Short Answer
Spectrophotometer: It is an optical instrument that measures intensity of light in relation to the wawelength.
Learning Materials
Features
Discover
Chapter 20: Q1P (page 525)
Describe the role of each component of the spectrophotometer.
Spectrophotometer: It is an optical instrument that measures intensity of light in relation to the wawelength.
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) The true absorbance of a sample is 1.500, but stray light reaches the detector. Find the apparent transmittance and apparent absorbance of the sample. (b) How much stray light can be tolerated if the absorbance error is not to exceedat a true absorbance of? (c) A research-quality spectrophotometer has a stray light level of ,at. What will be the maximum absorbance error for a sample with a true absorbance of 2? Of 3?
(a) In the cavity ring-down measurement at the opening of this chapter, absorbance is given by whereis the length of the triangular path in the cavity, Cis the speed of light, Tis the ring-down lifetime with sample in the cavity, and is the ring-down lifetime with no sample in the cavity. Ring-down lifetime is obtained by fitting the observed ring-down signal intensityto an exponential decay of the form , whereis the initial intensity and t is time. A measurement ofis made at a wavelength absorbed by the molecule. The ring-down lifetime for 21.0-cm-1 along empty cavity is andfor a cavity containing.role="math" localid="1664865078479" Find the absorbance ofat this wavelength.
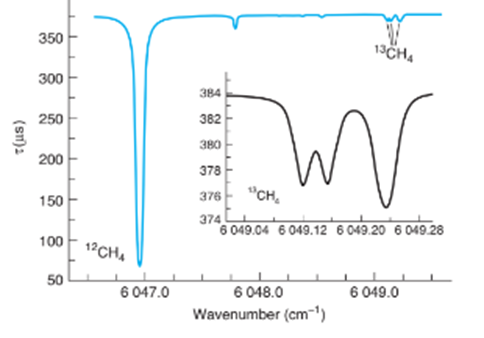
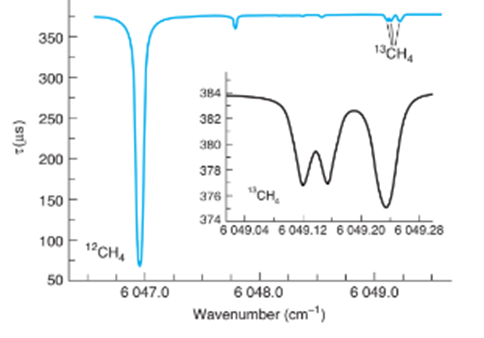
(b) The ring-down spectrum below arises from and from of methane in outdoor air at 0.13. The spectrum arises from individual rotational transitions of the ground vibrational state to a second excited C-H)vibrational state of the molecule. (i) Explain what quantity is plotted on the ordinate ( Y-axis). (ii) The peak foris at. What is the wavelength of this peak in? What is the name of the spectral region where this peak is found?
Describe three general types of noise that have a different dependence on frequency. Give an example of the source of each kind of noise.
The interferometer mirror of a Fourier transform infrared spectrophotometer travels pm1 cm .
(a) How many centimeters is the maximum retardation, ?
(b) State what is meant by resolution.
(c) What is the approximate resolution of the instrument?
(d) At what retardation interval, , must the interferogram be sampled (converted into digital form) to cover a spectral range of 0 to 2000 cm-1 ?
Find the minimum anglefor total reflection in the optical fiber in Figure 20-21 if the index of refraction of the cladding is 1.400 and the index of refraction of the core is
(a) 1.600 or
(b) 1.800.
What do you think about this solution?
We value your feedback to improve our textbook solutions.