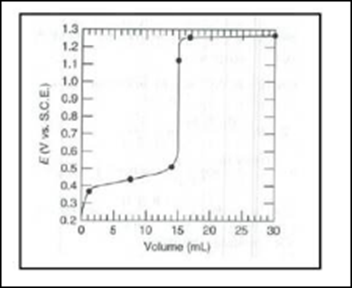
The potential vs. S.C.E. at titrant quantities of a particular volume must be computed, and a titration curve created.
Potential of the electrode (E): Electrode potential is the electromotive force between two electrodes. Two electrodes make up a cell: one is a standard electrode (such as calomel electrode or standard hydrogen electrode) and the other is a provided electrode.
Where,
is the electrode's potential when it's linked to the positive terminal
is the electrode's potential when it's linked to the negative terminal
Titration:
At 15.0 mL, the equivalency point is reached.
Prior to the moment of equivalency,
Use the electrode potential equation at the equivalence point,
After the equivalence point,
titration curve must be drawn