Chapter 5: Q24P (page 116)
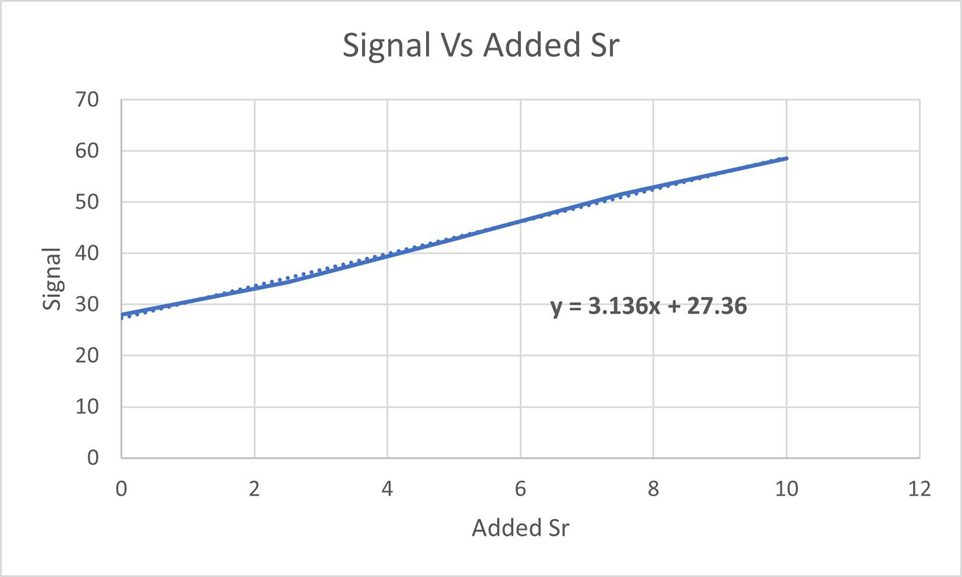
Standard addition graph. Tooth enamel consists mainly of the mineral calcium hydroxyapatite,. Trace elements in teeth of archeological specimens provide anthropologists with clues about diet and diseases of ancient people. Students at Hamline University used atomic absorption spectroscopy to measure strontium in enamel from extracted wisdom teeth. Solutions were prepared with a constant total volume of containing role="math" localid="1667792217398" of dissolved tooth enamel plus variable concentrations of added Sr.
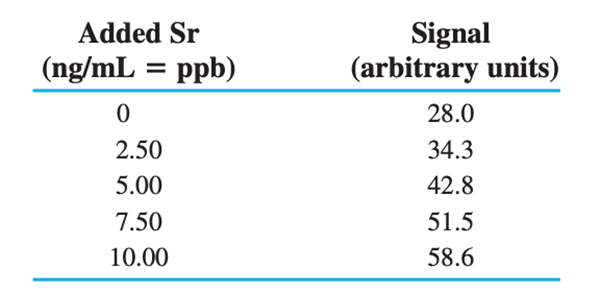
(a) Find the concentration of Sr and its uncertainty in therole="math" localid="1667792593357" sample solution in parts per billion =.
(b) Find the concentration of Sr in tooth enamel in parts per million =.
(c) If the standard addition intercept is the major source of uncertainty, find the uncertainty in the concentration of Sr in tooth enamel in parts per million.
(d) Find the 95%confidence interval for Sr in tooth enamel.
Short Answer
- Concentration, ; Uncertainty,.
- Concentration of Sr in tooth enamel =.
- The uncertainty in the concentration of Sr in tooth enamel= .
- confidence interval =.