Chapter 5: Q19P (page 115)
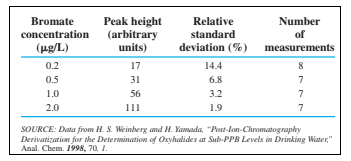
Detection limit. A sensitive chromatographic method was developed to measure sub-part-per-billion levels of the disinfectant by-products iodate, chlorite, and bromatein drinking water. As the oxyhalides emerge from the column, they absorption at. For example, each mole of bromate makes by the reaction
Bromate near its detection limit gave the following chromatographic peak heights and standard deviations (s). For each concentration, estimate the detection limit. Find the mean detection limit. The blank is 0 because chromatographic peak height is measured from the baseline adjacent to the peak. Because blank =0, relative standard deviation applies to both peak height and concentration, which are proportional to each other. Detection limit is 3 s for peak height or concentration.
Short Answer
The required concentration detection limit and mean CDL is


