Chapter 25: QEE (page 706)
Question:The graph shows retention data from asilica column with an acetonitrile/water mobile phase.
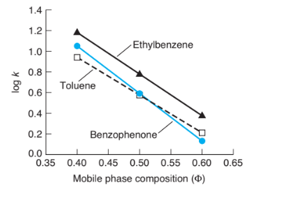
(a) What mobile phase composition provides greatest retentionfor the components? Least retention? Coelution of two components?
(b) Predict the retention time of each peak at . Draw a chromatogram (a "stick diagram" representing each peak as a vertical line) of the separation at each mobile phase composition.
(c) Wouldacetonitrile yield adequate resolution?
(d) Assuming Gaussian peaks, does the separation atacetonitrile have the attributes of a good separation?
Short Answer
a) The greatest and least retention of components in the mobile phase in reversed phase column chromatography, increasing the amount of organic solvent increases the mobile phase strength.
Acetonitrile at yields the longest retention time. The retention time is the shortest at .
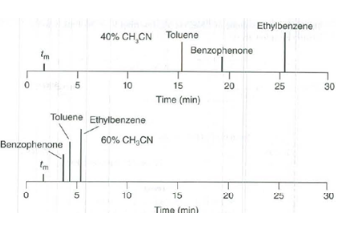
Toluene and benzophenone are co-elutes at acetonitrile.
b) The chromatogram:
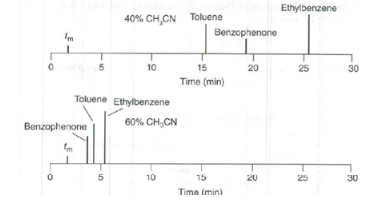
c) acetonitrile yield resolution.
d) The separation of peaks can be done by acetonitrile and it has additional plus point of separation time less than six minutes.