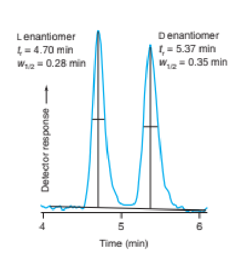
Chapter 25: Q20P (page 709)
Suppose that an HPLC column produces Gaussian peaks. The detector measures absorbance at 254 nm. A sample containing equal moles of compounds A and B was injected into the column. Compound A has a height h=128mm and a half-width mm. Compound B has mm. What is the height of peak B in millimeters?
Short Answer
The height of peak B in millimeters