Chapter 25: Q13P (page 707)
The figure shows the separation of two enantiomers on a chiral stationary phase.
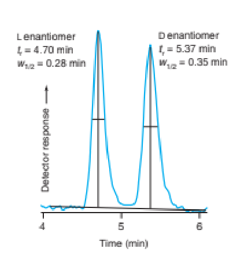
Sepation of enantiomers of Ritalin by HPLC with a chiral stationary phase.[Data from R.Bakhitar,L.Ramos,and F.L.S.Tse, “Quantification of methlylphenidate in plasma using chiral Liquid-chromatography/Tandem mass spectrometry: Application to Toxicokinetric studies,”Anal Chim Acta 2002,469,261.]
(a)From and find N for each peak.
(b) Fromandfind the resolution.
(c)Givenmin, use Equationwith the average N to predict the resolution.
Short Answer
The separation of two enantiomers on a chiral stationary phase.

