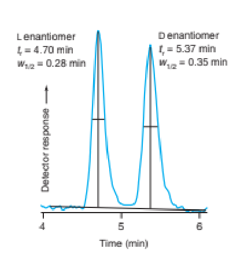
Chapter 25: Q40P (page 711)
Use Figure 25-30 for the following questions:
(a) What pH would be best for the separation of benzoic acid, 4-nitrophenol, and 3-methylbenzoic acid?
(b) What pH would be best for the separation of benzoic acid, 3-methylbenzoic acid, and 4-methylaniline?
(c) What pH would be best for separation of 4-nitrophenol, 4-methylaniline, and codeine on a typical C18-silica column?
Short Answer
(a).The value of pH = 2.0 .
(b).
(c).