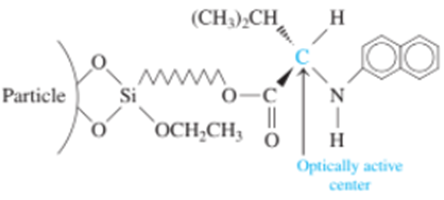
Chapter 25: Q8P (page 707)
a) UHPLC can provide exquisite resolution when run slowly on long columns or rapid separation with reasonaple resolution if short columns are run fast. the drug acetaminophern run on a UHPLC column has a retention time of min and a width at half-height of .Find the plate number and plate height. How many diameter particles places placed dide-by are equal to one theoretical plate?
b) From figure,we expect an optimum plate height of how many particles placed side-by- side are equal to one theroretical plate? Do you think the column in (a) is being run for maximum resolution or maximum speed?
Short Answer
UHPLC can provide exquisite resolution when run slowly on long columns.
how many particles placed side-by- side are equal to one theoretical plate.
particle/plate
b) particle/plate