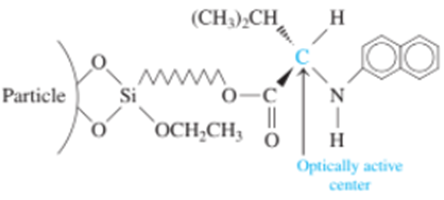
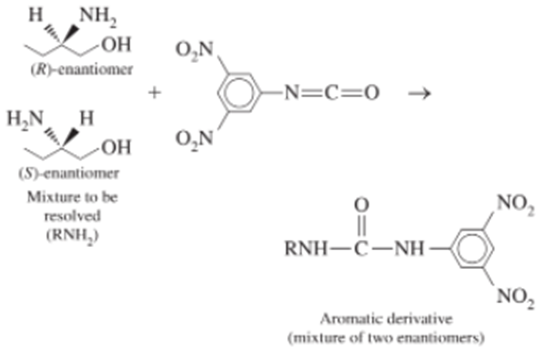
Chapter 25: Q33P (page 710)
(a) use figure 25-28a select a tetrahydrofurn/water mobile phase strength of equivalent strength to 80%methanol.
(b)Describe how to prepare 1 L of this tetrahydrofurn mobile phase.
(c)what limitation would be imposed by the use tetrahydrofurn.
Short Answer
a) 53%
b)To prepare 1 L of this tetrahydrofuran mobile phase (53 %), we need to mix 530 mL of tetrahydrofuran with 470 mL of water.
c) The limitations that would be imposed by the use of tetrahydrofuran is that it absorbs UV radiation and attacks polyether, ether, ketone and plastic components.