Chapter 18: Q9 P (page 456)
18-9 Why does a compound whose visible absorption maximum is at 480 nm (blue-green) appear to be red?
Short Answer
The Transmitted light will be complement of the color of absorption at 480 nm.
Learning Materials
Features
Discover
Chapter 18: Q9 P (page 456)
18-9 Why does a compound whose visible absorption maximum is at 480 nm (blue-green) appear to be red?
The Transmitted light will be complement of the color of absorption at 480 nm.
All the tools & learning materials you need for study success - in one app.
Get started for free
In formaldehyde, the transition occurs at 397 nm,and the transition comes at 355 nm. What is the difference in energy (kJ/mol) between the andstates? This difference is due to the different electron spins in the two states.
(a) How much energy (in kilojoules) is carried by one mole of photons of red light with= 650 nm?
(b) How many kilojoules are carried by one mole of photons of violet light with= 400 nm?
You have been sent to India to investigate the occurrence ofgoiter disease attributed to iodine deficiency. As part of your investigation, you must make field measurements of traces of iodide in groundwater. The procedure is to into an intensely colored complex with the dye brilliant green inthe organic solvent toluene.
(a)A solution of the colored complex exhibited an absorbance of 0.267 at 635nmin a 1.00 - cm cuvet. A blank solution made from distilled water in place of groundwater had an absorbance of 0.019 . Find the molar absorptivity of the coloredcomplex.
(b) The absorbance of an unknown solution prepared from groundwater was0.175. Find the concentration of the unknown.
If a sample for spectrophotometric analysis is placed in a 10 - cmcell, the absorbance will be 10times greater than the absorbance in a 1 - cmcell. Will the absorbance of the reagent-blank solution also be increased by a factor of 10?
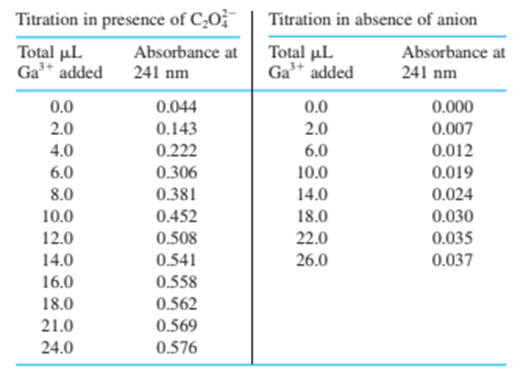
18-27. The iron-binding site of transferrin in Figure 18-8 can accommodate certain other metal ions besides Fe and certain other anions besidesData are given in the table for the titration of transferrin ( 3.57 mg in 2.00 mL ) with 6.64 mM solution in the presence of the anion oxalate, , and in the absence of a suitable anion. Prepare a graph similar to Figure 18-11, showing both sets of data. Indicate the theoretical equivalence point for the binding of one and two ions per molecule of protein and the observed end point. How many ions are bound to transferrin in the presence and in the absence of oxalate?
What do you think about this solution?
We value your feedback to improve our textbook solutions.